On-campus, T. cruzi screening is routinely performed to patients in Olive View-UCLA Medical Center who present with:
- cardiomyopahty
- abnormal electric conduction in ECG
- pregnancy
- newborns of Chagas positive mothers
Treatment in our center is offered once diagnosis is confirmed through the CDC. Currently only two drugs, Nifurtimox and Benznidazole, are available to use in the United States but, unfortunately none is FDA approved.
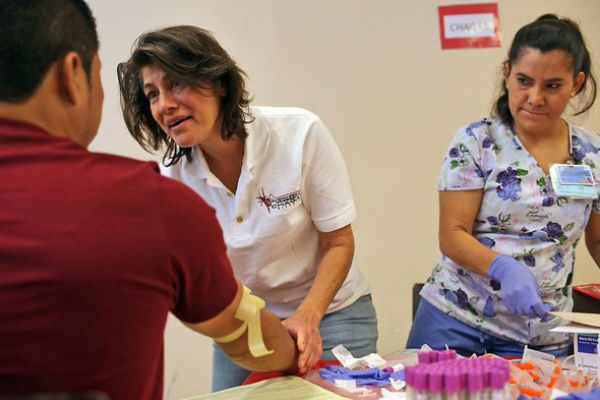
Once or twice a month, volunteers from our center go out to the community to churches, parks, civic centers to offer free Chagas screening at health fair events.
At the Center we are aware that Chagas patients are across the US and it is hard to travel to receive treatment. We offer guidance to physicians and health care providers from other counties and states so they can provide treatment to their patients
We are attempting to have the Department of Health Services (DHS) adopt screening for Chagas Disease in all of its facilities including 4 acute care hospital based primary care clinics and 19 ambulatory care clinics.
Towards this goal the laboratory at Olive View-UCLA Medical Center (OV) will become part of our Center of Excellence and will do the initial testing for CD at the hospital. All other DHS facilities will send blood samples to us for testing. Confirmatory testing will still be done at the CDC until the time that the United States Federal Drug Administration approves the drugs here for us. We are making good progress in this venue.
There are 2 million Latin American immigrants in LA County that are at risk of Chagas Disease. Many get their health care needs met by DHS. Just like blood pressure and blood sugar we would like to have a Chagas titers checked once in these clinical settings.
We are hopeful that within the next 2 years screening for Chagas Disease will be done at all DHS facilities. We will always continue our grass roots efforts to provide screening to those at risk who don’t have access to medical care.